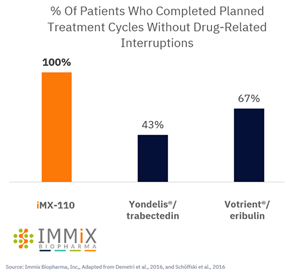
- 100% of patients treated with IMX-110 completed planned treatment cycles without drug-related interruptions in its ongoing Phase 1b/2a clinical trial
- IMX-110 is in clinical development as a monotherapy for soft tissue sarcoma (STS), a
$3 billion market expected to grow to$6.5 billion by 2030
A chart accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/b83a7ede-262b-4777-a2f9-beb13ee761bc
“With standard treatments widely used today, cancer patients face a long list of drug-related debilitating side-effects that cause treatment delays, dose reductions, or dose interruptions due to toxicity, interfering with the ability to treat them effectively,” said
The
About
Forward Looking Statements
This press release contains “forward-looking statements” Forward-looking statements reflect our current view about future events. When used in this press release, the words “anticipate,” “believe,” “estimate,” “expect,” “future,” “intend,” “plan,” or the negative of these terms and similar expressions, as they relate to us or our management, identify forward-looking statements. Such statements, include, but are not limited to, statements contained in this press release relating to our business strategy, our future operating results and liquidity and capital resources outlook. Forward-looking statements are based on our current expectations and assumptions regarding our business, the economy and other future conditions. Because forward–looking statements relate to the future, they are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict. Our actual results may differ materially from those contemplated by the forward-looking statements. They are neither statements of historical fact nor guarantees of assurance of future performance. We caution you therefore against relying on any of these forward-looking statements. Important factors that could cause actual results to differ materially from those in the forward-looking statements include, without limitation, our ability to raise capital to fund continuing operations; our ability to protect our intellectual property rights; the impact of any infringement actions or other litigation brought against us; competition from other providers and products; our ability to develop and commercialize products and services; changes in government regulation; our ability to complete capital raising transactions; and other factors relating to our industry, our operations and results of operations. Actual results may differ significantly from those anticipated, believed, estimated, expected, intended or planned.
Factors or events that could cause our actual results to differ may emerge from time to time, and it is not possible for us to predict all of them. We cannot guarantee future results, levels of activity, performance or achievements. The Company assumes no obligation to update any forward-looking statements in order to reflect any event or circumstance that may arise after the date of this release
Contacts
Chief Financial Officer
ir@immixbio.com
+1 (888) 958-1084

![]()
100% of Patients On IMX-110 Completed Planned Treatment Cycles
Immix Biopharma, Inc. (NASDAQ:IMMX)
2022 GlobeNewswire, Inc., source







