Bexotegrast demonstrated statistically significant increase in FVC at 4, 8 and 12 weeks of treatment, outperforming lower dose groups
No bexotegrast-treated patients experienced disease progression as defined by FVCpp decline of greater than or equal to 10%
Bexotegrast was well tolerated over 12 weeks of treatment with no drug-related severe or serious adverse events
Company to host webcast and conference call tomorrow,
The INTEGRIS-IPF Phase 2a trial is evaluating bexotegrast at once-daily doses of 40 mg, 80 mg, 160 mg, 320 mg or placebo for 12 weeks in 119 patients with IPF.
The 320 mg group enrolled 21 patients in the active arm and 8 patients in the placebo arm. Comparable to the lower dose groups, approximately 80% of all enrolled patients were on standard of care and were equally distributed between nintedanib and pirfenidone. The 320 mg group will continue until all patients have been treated for at least 24 weeks, with final data expected in the second quarter of 2023.
Bexotegrast 320 mg was Well Tolerated with No Drug-Related Severe or Serious Adverse Events
The primary endpoint of the INTEGRIS-IPF trial is the evaluation of the safety and tolerability of bexotegrast. The secondary endpoint is an assessment of its pharmacokinetics.
Bexotegrast was well tolerated at 320 mg over 12 weeks of treatment with no drug-related severe or serious adverse events (SAEs) reported. All drug-related adverse events were mild or moderate in severity.
Bexotegrast exhibited dose-proportional increases in plasma concentrations, consistent with prior studies.
Bexotegrast 320 mg Group Demonstrated Strong Treatment Effects on FVC, QLF and Profibrotic Biomarkers Versus Placebo over 12 Weeks
The exploratory efficacy endpoints of the INTEGRIS-IPF trial measured changes in FVC, high-resolution CT (HRCT)-based QLF, and profibrotic biomarkers over 12 weeks of treatment.
Figure 1. Change in FVC from Baseline of Bexotegrast 320 mg Over 12 Weeks in INTEGRIS-IPF; Mixed Model Repeat Measures Analysis – Modified Intent to Treat Population
A strong dose-dependent treatment effect was observed in the bexotegrast 320 mg dose group, with and without standard of care therapy. The bexotegrast 320 mg group demonstrated mean FVC increase of +29.5 mL relative to baseline at 12 weeks, versus a decline of 110.7 mL in the placebo group resulting in a 140 mL increase compared to placebo. Mean increases in FVC versus placebo reached statistical significance at all timepoints.
Figure 2. Proportion of Participants with FVCpp Decline ≥10% - Intent to Treat Population
A decline of ≥10% in FVCpp at 12 weeks has been associated with increased mortality in IPF patients over a two-year period.1,2 Over 12 weeks of treatment, no patients in the 320 mg group experienced ≥10% decline in FVCpp.
An increase in QLF score has been associated with worsening of pulmonary fibrosis.3 The mean percentage change in QLF at 12 weeks was 0.20% in the 320 mg group versus 1.46% in the placebo group.
Figure 3. Circulating PRO-C3 and Integrin beta-6 Biomarker Levels–Change from Baseline at 4- and 12-Weeks vs Placebo
PRO-C3, a serum biomarker of type III collagen synthesis, is elevated in patients with IPF and associated with progressive disease.4 The 320 mg group demonstrated a reduction in PRO-C3 at both 4 (p <0.01) and 12 weeks versus placebo.
Elevated integrin beta-6 plasma levels have been associated with ILD progression as defined by mortality, transplant or ≥ 10% relative reduction in FVC (mL) over 12 months.5 The 320 mg group demonstrated a reduction in integrin beta-6 at both 4 and 12 weeks (p <0.0001 at both timepoints) versus placebo.
These findings support a potential dose-dependent antifibrotic effect of bexotegrast, consistent with its mechanism of action and preclinical findings.
“Data from the INTEGRIS-IPF trial have far exceeded our expectations, supporting bexotegrast’s favorable safety profile and demonstrating a statistically significant treatment response on FVC at 320 mg,” said Éric Lefebvre, M.D., Chief Medical Officer at
“The statistically significant increase in mean FVC versus placebo seen throughout the 12-week treatment period of the INTEGRIS-IPF trial at 320 mg is unprecedented in clinical trials observed to date,” said
Bexotegrast Clinical Development Next Steps
Data from the 320 mg group of the INTEGRIS-IPF trial in patients treated for at least 24 weeks are expected in the second quarter of 2023. Pliant is planning to initiate a Phase 2b clinical trial of bexotegrast in mid-2023. Trial details will be shared closer to initiation.
INTEGRIS-IPF Multinational Phase 2 Trial of PLN-74809 (NCT04396756)
INTEGRIS-IPF is a Phase 2a, randomized, dose-ranging, double-blind, placebo-controlled trial evaluating the safety, tolerability, and pharmacokinetics of PLN-74809 administered in patients with IPF. Patients were enrolled in doses of 40 mg, 80 mg, 160 mg or 320 mg with a 3:1 randomization ratio (active:placebo) and stratification based on use of standard of care therapy. The primary endpoint is the evaluation of PLN-74809 safety and tolerability and the secondary endpoint is the assessment of pharmacokinetics across a dose range. Exploratory endpoints will measure change in Forced Vital Capacity (FVC), HRCT-based Quantitative Lung Fibrosis (QLF) score and selected biomarkers.
Background on Idiopathic Pulmonary Fibrosis
IPF is a chronic, progressive, fibrosing lung disease of unknown cause with few treatment options and a poor prognosis. Patients experience debilitating symptoms, including shortness of breath and difficulty performing daily activities, such as walking and talking. Currently, there is no pharmacological cure for IPF, with neither of the approved two therapies demonstrating an ability to stop the progression of IPF. Therefore, there is a high unmet need for new therapeutic options to address the symptoms and modify the disease progression of this grievous illness.
Conference Call and Webcast Information
The Company will host a conference call and webcast with a slide presentation tomorrow,
About
Forward-Looking Statements
Statements contained in this press release regarding matters that are not historical facts are "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as "may," "will," "expect," "anticipate," "estimate," "intend," and similar expressions (as well as other words or expressions referencing future events, conditions, or circumstances) are intended to identify forward-looking statements. These statements include those regarding the safety, tolerability, pharmacodynamics and therapeutic potential of PLN-74809; our plans for the future development of bexotegrast and PLN-101095; bexotegrast’s potential to become a treatment for IPF; the anticipated timing of data from the 320 mg group of the INTEGRIS-IPF Phase 2a trial in patients treated for at least 24 weeks; the timing and design of Pliant’s Phase 2b clinical trial of bexotegrast; discussions with regulatory authorities; and the efficacy and safety profile and potential of our product candidates. Because such statements deal with future events and are based on our current expectations, they are subject to various risks and uncertainties and actual results, performance or achievements of
Investor and Media Contact:
Vice President, Investor Relations and Corporate Communications
ir@pliantrx.com
1 Paterniti MO et al. Ann Am Thorac Soc. 2017 Sep 14(9):1395-1402.
2 Khan FA et al. Am J Respir Crit Care Med. 2022 Apr 15;205(8):936-948.
3 Kim GHJ et al. Ther Adv Respir Dis. 2021 Jan (15): 1–11.
4 Organ LA et al. Respir Res. 2019 Jul 12;20(1):148.
5 Bowman WS et al. Lancet Respir Med. 2022 Jun;10(6):593-602.
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/792e83b2-2e65-4312-a1a3-91a17bc67977
https://www.globenewswire.com/NewsRoom/AttachmentNg/8f751352-973a-43ba-9bd9-0ac2ea5dce04
https://www.globenewswire.com/NewsRoom/AttachmentNg/b67c8d9d-c550-42ca-a52b-f7a52a0a3749


Figure 1.
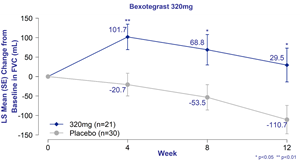
Change in FVC from Baseline of Bexotegrast 320 mg Over 12 Weeks in INTEGRIS-IPF; Mixed Model Repeat Measures Analysis – Modified Intent to Treat Population
Figure 2.
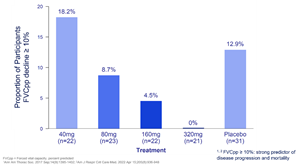
Proportion of Participants with FVCpp Decline ≥10% - Intent to Treat Population
Figure 3.

Circulating PRO-C3 and Integrin beta-6 Biomarker Levels– Change from Baseline at 4- and 12-Weeks vs Placebo
2023 GlobeNewswire, Inc., source







