- TCR-T Library Phase 1/2 trial achieved an 87% disease control rate of eight evaluable patients with metastatic, refractory solid tumors; TCR-T cell therapy was well tolerated in all treated patients
- hunTR® discovered new HLA class I and class II restricted TCRs that recognize driver mutations, including KRAS and TP53
- Company continues to explore potential strategic alternatives; cost-savings measures expected to extend cash runaway into the second quarter of 2024
Operational & Corporate Update
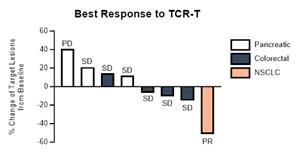
Clinical Data from TCR-T Library Phase 1/2 Trial: Eight patients were treated and evaluated in the Company’s TCR-T Library Phase 1/2 Trial. Patients with pancreatic (3), colorectal (4) and non-small cell lung cancer (1) were treated, with certain of the pancreatic and colorectal patients also having lung metastases. Overall, the trial showed the Company’s T cells were well-tolerated in all evaluable participants with no dose-limiting toxicities (DLTs) and no immune effector cell-associated neurotoxicity syndrome (ICANS) were observed. All cytokine release syndrome (CRS) events were within grades 1-3 and were self-limiting or resolved with standard clinical management and, in some cases, a single dose of tocilizumab.
One patient with non-small cell lung cancer (NSCLC) achieved an objective partial response with six months progression-free survival. Six other patients achieved best overall response of stable disease. The total overall response rate was 13% and disease control rate was 87% in evaluable patients with advanced, metastatic, refractory solid tumors (see attached figure). Disease control was measured by objective responses and stable disease. Increased secretion of interferon-gamma relative to baseline was detected in all patients’ serum post-cell transfer suggesting recognition of the tumor by the infused TCR-T cells. Persistence of TCR-T cells in peripheral blood was detected in all evaluable patients at their last follow-up, including up to six months in one patient. Infiltration of TCR-T cells into the tumor was also detected in three samples where a fresh biopsy was collected suggesting homing to the tumor microenvironment. All patients have progressed or withdrawn from the trial and long-term follow-up is ongoing for a subset of patients with no further intervention per the treatment protocol. This trial established proof-of-concept that Sleeping Beauty TCR-T cells can result in objective clinical responses and recognize established tumors in vivo.
Despite the encouraging TCR-T Library Phase 1/2 Trial data, based on the substantial cost to continue development and the current financing environment, Alaunos announced in
hunTR® TCR Discovery Platform Identifies Proprietary TCRs: Alaunos has discovered multiple proprietary TCRs targeting driver mutations through its hunTR® TCR discovery platform. In addition to TCRs that recognize KRAS and TP53 mutations similar to those licensed from the
Strategic Alternatives: The Company continues to explore strategic alternatives, which may include but are not limited to, an acquisition, merger, reverse merger, sale of assets, strategic partnerships, capital raises or other transactions. In connection with the strategic reprioritization, the Company has reduced its workforce by approximately 80% to date in order to streamline the organization and to maximize its cash runway.
Third Quarter Ended
Collaboration Revenue: Collaboration revenue was
Research and Development Expenses: Research and development expenses were
General and Administrative Expenses: General and administrative expenses were
Restructuring Costs: Restructuring costs were
Property and Equipment and Right-of-Use Asset Impairment: Property and equipment and right-of-use asset impairment charges were
Net Loss: Net loss was
Cash, Cash Equivalents and Restricted Cash: As of
About
Forward-Looking Statements Disclaimer
This press release contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts, and in some cases can be identified by terms such as “may,” “will,” “could,” “expects,” “plans,” “anticipates,” “believes” or other words or terms of similar meaning. These statements include, but are not limited to, statements regarding the Company’s ability to successfully implement its strategic reprioritization or realize any or all of the anticipated benefits once implemented and its ability to successfully consummate any strategic transactions; the completion and impact of the reduction in workforce; the planned renewed focus on the hunTR® TCR discovery platform and its success, including its ability to discover additional TCRs and the ability to monetize any newly discovered TCRs; the wind down of the TCR-T Phase 1/2 Library trial; the Company’s expected cash runway; and the results and potential of the TCR-T Phase 1/2 Library trial and the hunTR® discovery platform. Although the management team of Alaunos believes that the expectations reflected in such forward-looking statements are reasonable, investors are cautioned that forward-looking information and statements are subject to various risks and uncertainties, many of which are difficult to predict and generally beyond the control of Alaunos, that could cause actual results and developments to differ materially from those expressed in, or implied or projected by, the forward-looking information and statements. These risks and uncertainties include, among other things, risks relating to volatility and uncertainty in the capital markets for biotechnology and cell therapy companies; availability of suitable third parties with which to conduct contemplated strategic transactions; whether the Company will be able to pursue a strategic transaction, or whether any transaction, if pursued, will be completed successfully and on attractive terms or at all; whether our cash resources will be sufficient to fund the Company’s foreseeable and unforeseeable operating expenses and capital requirements; changes in the Company’s operating plans that may impact its cash expenditures; the uncertainties inherent in research and development, future clinical data and analysis; the risks associated with reductions in workforce, including reduced morale and attrition of additional employees necessary for the strategic reprioritization; the Company’s exclusive focus on its hunTR® TCR discovery platform; the strength and enforceability of Alaunos’ intellectual property rights; competition from other pharmaceutical and biotechnology companies; and the potential delisting of the Company’s common stock from the
Statement of Operations
(In thousands except per share data)
| For the Three Months Ended (Unaudited) | ||||||
| 2023 | 2022 | |||||
| Collaboration revenue | $ | - | $ | 2,911 | ||
| Operating expenses: | ||||||
| Research and development | $ | 3,656 | $ | 7,893 | ||
| General and administrative | 3,578 | 3,282 | ||||
| Restructuring costs | 419 | - | ||||
| Property and equipment and right-of-use asset impairment | 1,011 | - | ||||
| Total operating expenses | 8,664 | 11,175 | ||||
| Loss from operations | (8,664 | ) | (8,264 | ) | ||
| Interest expense | - | (841 | ) | |||
| Other income, net | 188 | 254 | ||||
| Net loss | (8,476 | ) | (8,851 | ) | ||
| Basic and diluted net loss per share | $ | (0.04 | ) | $ | (0.04 | ) |
| Weighted average common shares outstanding, basic and diluted | 240,046,026 | 215,098,995 | ||||
Selected Balance Sheet Data
(In thousands)
2023 (Unaudited) | 2022 (Audited) | |||||
| Cash and cash equivalents | $ | 11,944 | $ | 39,058 | ||
| Restricted cash | $ | - | $ | 13,938 | ||
| Working capital, excluding restricted cash | $ | 8,193 | $ | 15,695 | ||
| Total assets | $ | 19,440 | $ | 64,937 | ||
| Total stockholders’ equity | $ | 13,900 | $ | 38,555 | ||
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/004e98ab-3132-430a-8757-7e7dbd38f630

Investor Relations Contact: ir@alaunos.com
![]()
Best Response to TCR-T
Best Response to TCR-T
2023 GlobeNewswire, Inc., source

