- Cash runway extended into 2025 -
- Announced strategic program prioritization, expansion of manufacturing and plans to streamline business operations -
- Initiated Phase 1 clinical trial of RT-111, a RaniPill GO containing ustekinumab biosimilar CT-P43, with topline results expected in 1Q 2024 -
- Initiation of Phase 2 clinical trial of RT-102, a RaniPill GO containing teriparatide for osteoporosis, expected by the year-end 2023 -
- Announced RaniPill capsule was well-tolerated in 60-day, repeat oral-administration GLP safety study -
- Announced successful drug delivery with RaniPill HC high-capacity capsule in multiple preclinical studies with cumulative >90% success rate -
“In the third quarter, we made important decisions to drive forward our key programs to create long-term value for our shareholders and extend our cash runway into 2025. We announced a strategic prioritization of our RT-102 and RT-111 programs, as well as development of the RaniPill HC to be Phase 1 ready,” said
Third Quarter or Subsequent Highlights:
- Announced Strategic Program Prioritization, Expansion of Manufacturing and Plans to Streamline Business operations to Support Near-Term Value Drivers and Long-Term Growth of the RaniPill Technology Platform. The plans include strategic prioritization of its key development programs, RT-102, RT-111 and the RaniPill HC and expansion of its manufacturing footprint to support increased scale and partnerships, and cost reduction initiatives that align with Rani’s near-term goals. Development of RT-101 will be discontinued, while the development of RT-105 and RT-110 programs will be paused. In addition, Rani will reduce its workforce by approximately 25%. Anticipated cost savings are expected to support Rani’s operating plans into 2025.
- Completed 60-Day, Repeat Oral-Administration GLP Safety Study. In
October 2023 , Rani announced preclinical data from a 60-day, repeat oral-administration GLP safety study of the RaniPill capsule in healthy animals. The RaniPill capsule was well-tolerated with no treatment-related adverse events and all animals remained clinically healthy throughout the study. - Presented an Abstract on RT-102 at the 2023 Annual Meeting of the
American Society for Bone and Mineral Research . The abstract focused on the safe and reliable delivery of teriparatide with high bioavailability through daily administration of an oral robotic pill (RT-102) in female volunteers. - Initiated Phase 1 Clinical Trial of RT-111 (RaniPill Containing Ustekinumab Biosimilar, CT-P43). In
September 2023 , Rani announced the initiation of a Phase 1 clinical trial to evaluate the safety and tolerability of RT-111, an orally administered RaniPill GO capsule containing an ustekinumab biosimilar, CT-P43. Currently, ustekinumab is available only as a subcutaneous injection. In preclinical testing of RT-111 in animal models, the RaniPill delivered ustekinumab biosimilar orally with bioavailability comparable to subcutaneous injection. Topline results from this study are expected early in the first quarter of 2024. - Announced Successful Drug Delivery of High-Capacity Pill in Preclinical Studies. In September and
October 2023 , Rani announced results from multiple preclinical studies of the RaniPill HC, a version of the RaniPill capsule capable of delivering up to a 500%-plus higher drug payload than Rani’s existing oral biologics capsule. In such studies, the RaniPill HC achieved successful drug delivery with a cumulative >90% success rate.
Development Update
- Rani has completed two preclinical studies of the RaniPill HC with antibodies, adalimumab and an undisclosed interleukin (“Interleukin”). In the two antibody studies, the RaniPill HC achieved an oral delivery success rate of 100% (10/10). In one study, Rani tracked the serum concentrations of adalimumab, following the oral administration of the enteric-coated, RaniPill HC capsule containing 11mg of Humira (adalimumab) to four canine models. In the second study, Rani tracked the serum concentrations of the Interleukin, following the oral administration of the enteric-coated RaniPill HC capsule containing 16.5mg of Interleukin to six canine models. In both studies, the RaniPill HC was well-tolerated, all animals remained healthy throughout the study period with no clinical findings or adverse events, and all device remnants were excreted normally without sequelae.
- Comparing the pharmacokinetic results of 11mg of adalimumab delivered via the RaniPill HC (N=4) with historical pharmacokinetic data generated by Rani with 5mg of an adalimumab biosimilar (GP2017) delivered via subcutaneous injection (N=3), there is a higher estimated bioavailability of adalimumab delivered via the RaniPill HC relative to the subcutaneous injection route.
Adalimumab 11mg via RaniPill HC vs Adalimumab Biosimilar 5mg via Subcutaneous Injection
All Data are Means ± SE
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/17476063-074d-43a1-9815-963636ade293
Pharmacokinetics of Interleukin (16.5mg) Delivered Orally via RaniPill HC Capsules to Awake Canines (N=6)
All Data are Means ± SE
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/d742871b-dcba-4f9b-8afa-f7bf8e66783d
Preliminary preclinical testing supports the potential for the RaniPill HC to have high reliability and initial analysis of drug delivery via the RaniPill HC shows a potential for mimicking parenteral (subcutaneous) administration. Rani expects to continue preclinical testing of RaniPill HC to confirm the preliminary reliability rate and optimize device performance with a goal for the RaniPill HC to be ready to advance into the clinic in the second half of 2024.
Near-Term Milestone Expectations:
- Initiation of Phase 2 clinical trial of RT-102, a RaniPill GO containing teriparatide for osteoporosis, expected by year-end 2023.
- Topline results of Phase 1 clinical trial of RT-111, a RaniPill GO containing ustekinumab biosimilar CT-P43, expected in the first quarter of 2024.
- Development of RaniPill HC to be ready for potential Phase 1 clinical trials in the second half of 2024.
Third Quarter Financial Results:
- Cash, cash equivalents and marketable securities as of
September 30, 2023 , totaled$60.5 million , compared to cash, cash equivalents and marketable securities of$98.5 million as ofDecember 31, 2022 . Rani expects its cash, cash equivalents and marketable securities to be sufficient to fund its operations through at least the next twelve months. - Research and development expenses were
$11.2 million for the three months endedSeptember 30, 2023 , compared to$9.1 million for the three months endedSeptember 30, 2022 . The increase was primarily attributed to higher third-party services expense of$2.1 million due to pre-clinical and clinical development activities. - General and administrative expenses were
$6.6 million for the three months endedSeptember 30, 2023 , compared to$7.2 million for the three months endedSeptember 30, 2022 . The decrease was primarily attributed to third-party services of$0.7 million related to support for compliance with public company requirements. - Net loss for the three months ended
September 30, 2023 was$18.3 million , compared to$16.2 million for the comparable period in 2022, including stock-based compensation expense of$5.0 million for the three months endedSeptember 30, 2023 compared to$4.4 million for the comparable period in 2022.
About
Forward-Looking Statements
Statements contained in this press release regarding matters that are not historical facts are “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements include statements regarding, among other things, the advancement of Rani’s pipeline and RaniPill platform technology including the RaniPill HC, the potential of the RaniPill HC to deliver 500%-plus higher drug payload than Rani’s existing oral biologics capsule, the expected initiation of a Phase 2 clinical trial of RT-102 in 2023, the ability to confirm preliminary reliability and optimize performance of the RaniPill HC, the expected timing of topline results from the RT-111 Phase 1 clinical trial in the first quarter of 2024, the expected readiness of the RaniPill HC for clinical development in the second half of 2024, the ability of expanded manufacturing footpoint to support scaling of manufacturing and partnering, the ability to streamline its business operations and to realize the cost-savings contemplated by such streamlining of business operations, reduction in workforce and other initiatives announced by Rani, the potential for the RaniPill HC to have high reliability and mimic parenteral (subcutaneous) administration, the potential for partner opportunities with the RaniPill HC, customer acceptance of the RaniPill capsule technology, the potential benefits of the RaniPill capsule technology, cash sufficiency forecast, and Rani’s growth as a company. Because such statements are subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such forward-looking statements. Words such as “capable of,” “potential,” “expects,” “with a goal for,” “anticipate” and similar expressions are intended to identify forward-looking statements. These forward-looking statements are based upon Rani’s current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results could differ materially from those anticipated in such forward-looking statements as a result of various risks and uncertainties, which include, without limitation, risks and uncertainties associated with Rani’s business in general and the other risks described in Rani’s filings with the
Trademarks
Trade names, trademarks and service marks of other companies appearing in this press release are the property of their respective owners. Solely for convenience, the trademarks and trade names referred to in this press release appear without the ® and ™ symbols, but those references are not intended to indicate, in any way, that we will not assert, to the fullest extent under applicable law, our rights, or the right of the applicable licensor to these trademarks and tradenames.
Investor Contact:
investors@ranitherapeutics.com
Media Contact:
media@ranitherapeutics.com
Condensed Consolidated Balance Sheets
(In thousands, except par value)
| 2023 | 2022 | |||||||
| (Unaudited) | ||||||||
| Assets | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 4,972 | $ | 27,007 | ||||
| Marketable securities | 55,554 | 71,475 | ||||||
| Prepaid expenses and other current assets | 2,696 | 2,442 | ||||||
| Total current assets | 63,222 | 100,924 | ||||||
| Property and equipment, net | 6,255 | 6,038 | ||||||
| Operating lease right-of-use asset | 959 | 1,065 | ||||||
| Total assets | $ | 70,436 | $ | 108,027 | ||||
| Liabilities and Stockholders' Equity | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 1,405 | $ | 1,460 | ||||
| Accrued expenses and other current liabilities | 3,930 | 2,349 | ||||||
| Current portion of long-term debt | 1,222 | — | ||||||
| Current portion of operating lease liability | 788 | 1,006 | ||||||
| Total current liabilities | 7,345 | 4,815 | ||||||
| Long-term debt, less current portion | 28,101 | 29,149 | ||||||
| Operating lease liability, less current portion | 171 | 59 | ||||||
| Total liabilities | 35,617 | 34,023 | ||||||
| Stockholders' equity: | ||||||||
| Preferred stock, | — | — | ||||||
| Class A common stock, | 3 | 3 | ||||||
| Class B common stock, | 2 | 2 | ||||||
| Class C common stock, | — | — | ||||||
| Additional paid-in capital | 83,380 | 75,842 | ||||||
| Accumulated other comprehensive loss | (41 | ) | (73 | ) | ||||
| Accumulated deficit | (65,791 | ) | (38,919 | ) | ||||
| Total stockholders' equity attributable to | 17,553 | 36,855 | ||||||
| Non-controlling interest | 17,266 | 37,149 | ||||||
| Total stockholders' equity | 34,819 | 74,004 | ||||||
| Total liabilities and stockholders' equity | $ | 70,436 | $ | 108,027 | ||||
Condensed Consolidated Statements of Operations
(In thousands, except per share amounts)
(Unaudited)
| Three Months Ended | Nine Months Ended | |||||||||||||||
| 2023 | 2022 | 2023 | 2022 | |||||||||||||
| Operating expenses | ||||||||||||||||
| Research and development | $ | 11,220 | $ | 9,103 | $ | 32,018 | $ | 26,221 | ||||||||
| General and administrative | 6,635 | 7,239 | 20,647 | 19,748 | ||||||||||||
| Total operating expenses | $ | 17,855 | $ | 16,342 | $ | 52,665 | $ | 45,969 | ||||||||
| Loss from operations | (17,855 | ) | (16,342 | ) | (52,665 | ) | (45,969 | ) | ||||||||
| Other income (expense), net | ||||||||||||||||
| Interest income and other, net | 839 | 379 | 2,626 | 430 | ||||||||||||
| Interest expense and other, net | (1,316 | ) | (352 | ) | (3,789 | ) | (352 | ) | ||||||||
| Loss before income taxes | (18,332 | ) | (16,315 | ) | (53,828 | ) | (45,891 | ) | ||||||||
| Income tax expense | — | 107 | — | (111 | ) | |||||||||||
| Net loss | $ | (18,332 | ) | $ | (16,208 | ) | $ | (53,828 | ) | $ | (46,002 | ) | ||||
| Net loss attributable to non-controlling interest | (9,135 | ) | (8,253 | ) | (26,956 | ) | (24,200 | ) | ||||||||
| Net loss attributable to | $ | (9,197 | ) | $ | (7,955 | ) | $ | (26,872 | ) | $ | (21,802 | ) | ||||
| Net loss per Class A common share attributable to | $ | (0.36 | ) | $ | (0.33 | ) | $ | (1.06 | ) | $ | (0.93 | ) | ||||
| Weighted-average Class A common shares outstanding—basic and diluted | 25,552 | 24,468 | 25,380 | 23,449 | ||||||||||||


Adalimumab 11mg via RaniPill HC vs Adalimumab Biosimilar 5mg via Subcutaneous Injection
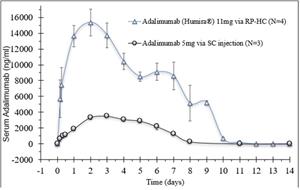
Adalimumab 11mg via RaniPill HC vs Adalimumab Biosimilar 5mg via Subcutaneous Injection
Pharmacokinetics of Interleukin (16.5mg) Delivered Orally via RaniPill HC Capsules to Awake Canines (N=6)
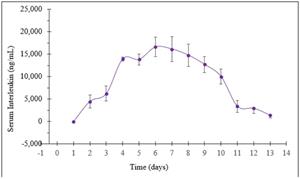
Pharmacokinetics of Interleukin (16.5mg) Delivered Orally via RaniPill HC Capsules to Awake Canines (N=6)
2023 GlobeNewswire, Inc., source

