PYX-201 Phase 1 trial clinical readout on track for fall of 2024
Executive Leadership Team expanded with the appointment of
PYX-106 Phase 1 trial clinical readout on track for 2H 2024
Expected cash runway into 2H 2026
PYX-201, a first-in-concept tumor stroma targeting antibody-drug conjugate (ADC) against the stromal Extradomain-B Fibronectin (EDB+FN) target, has dosed 42 patients in 8 cohorts since initiating the Phase 1 trial in
"Based on encouraging early responses with late-stage patients across multiple tumor types, we are actively studying dose ranges from 5.4 mg/kg to 8 mg/kg, refining our understanding of PYX-201's therapeutic window. We are on track to report the comprehensive dataset in the fall of 2024, and we look forward to the potential future robust development plan supported by our strong balance sheet," said
Another poster (Figure 2. Abstract #2908)
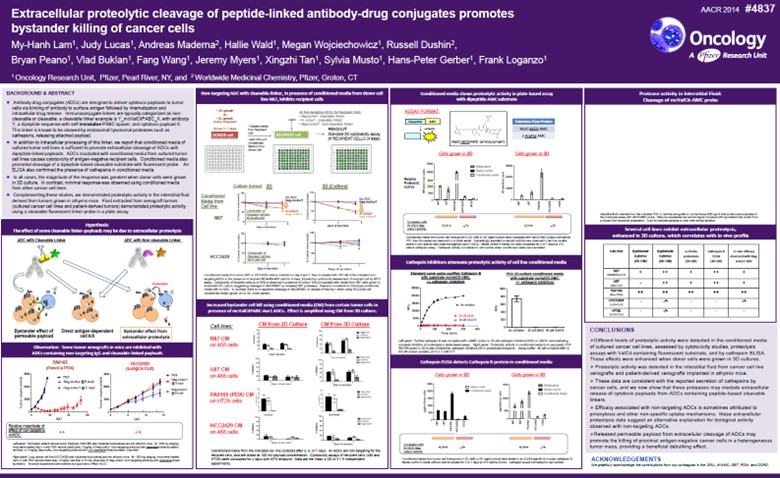
Given the observed anti-tumor activity for our non-internalizing mechanism of action,
In summary, Pyxis Oncology’s preclinical data provides insights into the mechanism associated with this novel agent observed across multiple solid tumors. PYX-201 has potential applications in both monotherapy and combination therapy and maintains a well-tolerated safety profile based on the lack of EDB+FN expression in healthy cells.
Concurrently,
In his role, Stephen will oversee
"We are thrilled to welcome Stephen to
"Joining
Stephen joins
Program and Corporate Updates
- PYX-201 in the PYX-201-101 trial: To date, 42 subjects have been dosed, and the Company will enroll an additional 16 patients.
Pyxis Oncology expects to report study results, including efficacy, safety, pharmacokinetics (PK), preclinical insights, further development plans, and the expected timing of the next anticipated milestones in the fall of 2024. - PYX-106 in the PYX-106-101 trial: This is a phase 1 trial focusing on NSCLC and other tumor types. Study dosing is ongoing, with 24 subjects dosed to date. Preliminary data are anticipated in 2H 2024.
- AACR Poster Presentations. Presented new PYX-201, PYX-106 and PYX-102 preclinical data at the AACR Annual Meeting.
- Expanded Executive Leadership Team with the appointment of
Stephen Worsley as Senior Vice President, Chief Business Officer.
Anticipated Upcoming Milestones
- PYX-201: Report preliminary Phase 1 data and PK/PD results in fall of 2024
- PYX-106: Report preliminary Phase 1 data and PK/PD results in 2H 2024, following the release of PYX-201 results
First Quarter 2024 Financial Results
- As of
March 31, 2024 ,Pyxis Oncology had cash and cash equivalents, including restricted cash, and short-term investments of$158.5 million . During Q1 2024, the Company raised gross proceeds of$10.8 million via an at-the-market (“ATM”) offering, completed a$50 million private placement, and sold the Company’s rights to royalties from the commercialization of Beovu® (brolucizumab-dbll) and another asset for a one-time payment of$8 million to Novartis.Pyxis Oncology expects to have the cash runway to fund operations into 2H 2026. - Revenues for the quarter ended
March 31, 2024 were$16.1 million , compared to$0 for the quarter endedMarch 31, 2023 . During the quarter, we entered into a settlement agreement with Novartis, pursuant to which we transferred our rights to future royalties on the net sales of Beovu® to Novartis for a one-time amount of$8.0 million and Novartis also agreed to forgo its right to reclaim royalties previously paid of$8.1 million to us and Apexigen. Both of these amounts were recognized as revenues, upon execution of the settlement agreement during the quarter endedMarch 31, 2024 . - Research and development expenses were
$13.0 million for the quarter endedMarch 31, 2024 , compared to$11.9 million for the quarter endedMarch 31, 2023 . The period-over-period increase was primarily due to increased clinical trial-related expenses for our ongoing Phase 1 clinical trials of PYX-201 and PYX-106. - General and administrative expenses were
$8.2 million for the quarter endedMarch 31, 2024 , compared to$9.1 million for the quarter endedMarch 31, 2023 . The period-over-period decline was primarily due to lower professional and consultant fees. - Net loss was
$3.3 million , or ($0.06 ) per common share, for the quarter endedMarch 31, 2024 , compared to$19.2 million , or ($0.54 ) per common share, for the quarter endedMarch 31, 2023 . Net losses for the quarters endedMarch 31, 2024 and 2023 included$4.3 million and$4.9 million , respectively, related to non-cash stock-based compensation expense. - As of
May 13, 2024 , the outstanding number of shares of Common Stock ofPyxis Oncology was 58,888,473.
About
Forward-Looking Statements
This press release contains forward-looking statements for the purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995 and other federal securities laws. These statements are often identified by the use of words such as “on track,” “anticipate,” “believe,” “can,” “continue,” “could,” “estimate,” “expect,” “intend,” “likely,” “may,” “might,” “objective,” “ongoing,” “plan,” “potential,” “predict,” “project,” “should,” “to be,” “will,” “would,” or the negative or plural of these words, or similar expressions or variations, although not all forward-looking statements contain these words. We cannot assure you that the events and circumstances reflected in the forward-looking statements will be achieved or occur and actual results could differ materially from those expressed or implied by these forward-looking statements. Factors that could cause or contribute to such differences include, but are not limited to, those identified herein, and those discussed in the section titled “Risk Factors” set forth in Part II, Item 1A. of the Company’s Annual Report on Form 10-K filed with
Pyxis Oncology Contact
CFO and COO
ir@pyxisoncology.com
FIGURES
Figure 1. AACR 2024 – Abstract Number: 742
PYX-201, a stroma-targeting ADC composed of an anti-EDB+FN antibody conjugated to Auristatin0101, demonstrates strong anti-tumor efficacy across multiple human cancer indications in pre-clinical PDX tumor models
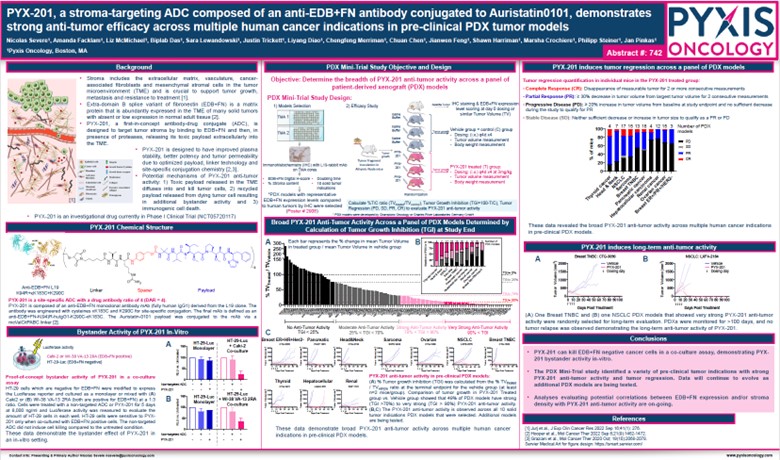
Figure 2. AACR 2024 – Abstract Number: 2908
PYX-201, EDB+FN is an attractive target in oncology: Insights from protein expression analysis of solid tumors
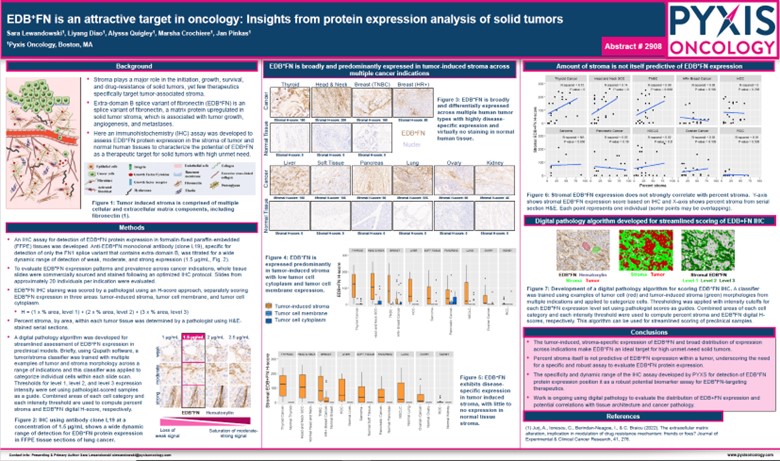
Figure 3. AACR 2014 – Abstract Number: 4837
Extracellular proteolytic cleavage of peptide-linked antibody-drug conjugates promotes bystander killing of cancer cells
---tables to follow---
Condensed Consolidated Statements of Operations and Comprehensive Loss (In thousands, except share and per share amounts) (Unaudited) | ||||||||
| Three Months Ended | ||||||||
| 2024 | 2023 | |||||||
| Revenues | ||||||||
| Royalty revenues | $ | 8,146 | $ | — | ||||
| Sale of royalty rights | 8,000 | — | ||||||
| Total revenues | 16,146 | — | ||||||
| Costs and operating expenses: | ||||||||
| Cost of revenues | 475 | — | ||||||
| Research and development | 13,029 | 11,901 | ||||||
| General and administrative | 8,247 | 9,053 | ||||||
| Total costs and operating expenses | 21,751 | 20,954 | ||||||
| Loss from operations | (5,605 | ) | (20,954 | ) | ||||
| Other income, net: | ||||||||
| Interest and investment income | 1,550 | 1,673 | ||||||
| Sublease income | 799 | 38 | ||||||
| Total other income, net | 2,349 | 1,711 | ||||||
| Net loss | $ | (3,256 | ) | $ | (19,243 | ) | ||
| Net loss per common share - basic and diluted | $ | (0.06 | ) | $ | (0.54 | ) | ||
| Weighted average shares of common stock outstanding - basic and diluted | 51,289,284 | 35,351,671 | ||||||
Condensed Consolidated Balance Sheets (In thousands, except share and per share amounts) (Unaudited) | ||||||||
| Assets | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 27,967 | $ | 9,664 | ||||
| Marketable debt securities, short-term | 129,060 | 109,634 | ||||||
| Restricted cash | 1,472 | 1,472 | ||||||
| Accounts receivable | 8,000 | — | ||||||
| Prepaid expenses and other current assets | 5,880 | 3,834 | ||||||
| Total current assets | 172,379 | 124,604 | ||||||
| Property and equipment, net | 11,333 | 11,872 | ||||||
| Intangible assets, net | 23,730 | 24,308 | ||||||
| Operating lease right-of-use assets | 12,778 | 12,942 | ||||||
| Total assets | $ | 220,220 | $ | 173,726 | ||||
| Liabilities and Stockholders’ Equity | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 2,293 | $ | 3,896 | ||||
| Accrued expenses and other current liabilities | 10,828 | 12,971 | ||||||
| Operating lease liabilities, current portion | 1,020 | 1,232 | ||||||
| Deferred revenues | — | 7,660 | ||||||
| Total current liabilities | 14,141 | 25,759 | ||||||
| Operating lease liabilities, net of current portion | 19,759 | 20,099 | ||||||
| Deferred tax liability, net | 2,164 | 2,164 | ||||||
| Total liabilities | 36,064 | 48,022 | ||||||
| Commitments and contingencies | ||||||||
| Stockholders’ equity: | ||||||||
| Preferred stock, par value | — | — | ||||||
| Common stock, | 59 | 45 | ||||||
| Additional paid-in capital | 473,638 | 411,821 | ||||||
| Accumulated other comprehensive (loss) income | (60 | ) | 63 | |||||
| Accumulated deficit | (289,481 | ) | (286,225 | ) | ||||
| Total stockholders’ equity | 184,156 | 125,704 | ||||||
| Total liabilities and stockholders’ equity | $ | 220,220 | $ | 173,726 | ||||
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/ab8e54f0-f5b9-493c-9c94-0623bf1b0e93
https://www.globenewswire.com/NewsRoom/AttachmentNg/76e0d9fd-3532-4460-9926-a6bec1ce56c0
https://www.globenewswire.com/NewsRoom/AttachmentNg/89db2061-133e-43a4-bc33-3cc7780ba80e


Figure 1

AACR 2024 – Abstract Number: 742
Figure 2

AACR 2024 – Abstract Number: 2908
Figure 3

AACR 2014 – Abstract Number: 4837
2024 GlobeNewswire, Inc., source







