- Lab-quality results for both COVID-19 and Flu now available at home from one single test
- COVID-19 and Flu have similar symptoms but different treatments, requiring a fast, differential test to access time-sensitive prescription treatment options
- Lucira’s COVID-19 & Flu Home Test demonstrated similar performance for COVID-19, Flu A and Flu B in head-to-head comparison study with highly sensitive lab-based PCR tests
- Lucira seeks a strategic or financial partner for resumption of manufacturing and development of additional home diagnostic products
Lucira believes that a single test for both COVID-19 and Flu can be a powerful tool given how similar the two viruses appear. “COVID-19 and Flu look the same, feel the same, spread the same and, unfortunately, can still kill the same,” underscored Dr.
“Our small but experienced team at Lucira has again demonstrated the versatility of our technology platform by introducing this first-of-its-kind innovation in clinically relevant at-home diagnostics to the marketplace. The authorization of the COVID-19 & Flu Home Test with OTC label is another example of how Lucira is helping to transform the future promise of home-based, fast, accurate diagnostics into a reality,” said
“We regret that we had no option but to file for Chapter 11 bankruptcy and that this occurred days before we received regulatory authorization,” added
Lucira is seeking a strategic or financial partner for the resumption of manufacturing and development of additional home diagnostic products.
The Company believes that its at-home, accurate diagnostic products are a critical third leg of the stool that also includes telehealth and home prescription delivery for the future of safe, home-based healthcare. Lucira’s expectation is that as accurate home diagnostics become prevalent, suffering and disease spread can be reduced, and that all constituents, including the public health, the healthcare value chain as well as the cost of healthcare can benefit. In the end, consumers may think of visiting a healthcare office for infectious disease diagnoses and treatment as being as antiquated as going to a video rental store to get a movie to watch: as a thing of the past.
For more information on the Lucira COVID-19 & Flu Home Test, please visit
www.lucirahealth.com.
About the Lucira COVID-19 & Flu Home Test
The Lucira COVID-19 & Flu Home Test is not an antigen test. It is a molecular, nucleic acid amplification (NAAT) test that utilizes the same platform and device design as both of Lucira's commercialized FDA authorized COVID-19 tests to provide independent diagnoses for COVID-19, Flu A and Flu B. The lab-quality single-use test fits in the palm of your hand, runs on 2 AA batteries, and with one shallow nasal swab provides a positive or negative result for COVID-19, Flu A and Flu B in 30 minutes or less. Each Lucira test contains everything needed to run a single test. There is no separate reader or instrument to purchase and maintain.
About
Lucira is a medical technology company focused on the development and commercialization of innovative infectious disease tests to make lab-quality diagnostics more accessible. Lucira designed its test platform to provide accurate, reliable, lab-quality test results anywhere and at any time. Beyond its already commercialized molecular COVID-19 and COVID-19 & Flu tests, Lucira is working on new diagnostic tests for respiratory infections and other categories including women’s health and sexually transmitted infections (STIs). For more information, visit www.lucirahealth.com.
Forward Looking Statements
Statements contained in this press release regarding matters that are not historical facts are "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Because such statements are subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such forward-looking statements. Words such as “can,” “will,” “seek,” “believe,” “should,” “future,” “look to,” “continue” and similar expressions are intended to identify forward-looking statements. These forward-looking statements, including but not limited to, our belief that a single test for both COVID-19 and Flu will be a powerful tool; at-home molecular test helping people know how to keep their families safe and seek appropriate treatment when they’re ill; and how OTC authorization is helping Lucira transform the future promise of home-based, fast, accurate diagnostics into a reality; the role the new test can play in future respiratory seasons; Lucira’s seeking a strategic or financial partner for the resumption of manufacturing and development of additional diagnostic products and making products available in retail and online and bringing additional diagnostic products to market; Lucira’s work on new diagnostic tests for respiratory infections, women’s health and STIs; and Lucira’s expectation that accurate home diagnostics will become prevalent and reduce suffering and disease spread, thereby benefiting all constituents, are based upon Lucira’s current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results could differ materially from those anticipated in such forward-looking statements as a result of various risks and uncertainties. These risks and uncertainties are described more fully in the "Risk Factors" section and elsewhere in Lucira’s filings with the
Media Contact:
media@lucirahealth.com
Investor Contact:
Email: gwiener@GWCco.com
Photos accompanying this announcement are available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/2f5ac3ad-e26a-4b70-bee2-2f20dc498aee
https://www.globenewswire.com/NewsRoom/AttachmentNg/a1fefbb4-2962-4057-8bb4-abe6118db212
https://www.globenewswire.com/NewsRoom/AttachmentNg/a2dc1f95-4bdc-4e89-9754-7dc8037c568d

![]()
Lucira COVID-19 & Flu Home Test – The First & Only Combination Test for OTC Use At Home
Is it Covid, or the Flu? It can be hard to tell which virus you have based on symptoms alone, but you need to know to get the right treatment. Now, with the Lucira COVID-19 & Flu Home Test, Americans can for the first time accurately self-diagnose whether it is Covid or the Flu with molecular level accuracy in 30 minutes or less at home.
Lucira COVID-19 & Flu Home Test – The First OTC Test to Diagnose Flu at Home in History
Americans have never before been able to accurately self-diagnose Flu at-home, until today. The Lucira COVID-19 & Flu Home Test is the only combination test to diagnose Covid and Flu at home, and is the first at-home test to ever allow self-diagnosis of Flu. Now you can tell whether it is Covid or the Flu with molecular level accuracy in 30 minutes or less at home.
Lucira COVID-19 & Flu Home Test – All You Need to Answer “Is it Covid or the Flu?”
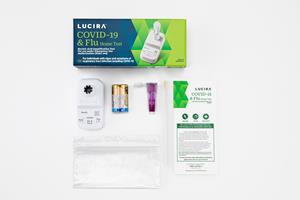
Flu and Covid continue to co-circulate, and it can be hard to tell which virus you have based on symptoms alone. The Lucira COVID-19 & Flu Home Test has everything you need to accurately diagnose whether it is Covid or the Flu in 30 minutes or less in the comfort of your home.
2023 GlobeNewswire, Inc., source

